If your semen analysis came back showing low sperm motility, you're probably feeling a mix of worry and confusion. What does it actually mean? Is it fixable? And does it mean you can't have children naturally?
Here's the reassuring truth: low sperm motility—medically known as asthenozoospermia—is one of the most common findings on a semen analysis, and in many cases, it can be improved. It's also worth knowing that asthenozoospermia is diagnosed in up to 82% of infertile men to varying degrees, so you are far from alone.
This guide explains exactly what low motility means, walks through every known cause from varicocele to lifestyle factors, and covers what the research actually says about treatments and supplements—so you can make informed decisions about your next steps.
Also see: Understanding Semen Analysis Results for a full breakdown of every parameter on your report.
What Is Sperm Motility and Why Does It Matter?
Sperm motility is simply the ability of sperm to move. But not all movement is equal. For natural conception, sperm need to do far more than just wiggle—they need to swim actively forward through the cervix, uterus, and fallopian tubes to reach and fertilise the egg. That's a journey of roughly 15–18 centimetres, which for a cell only 50 micrometres long is the equivalent of a human swimming several kilometres.
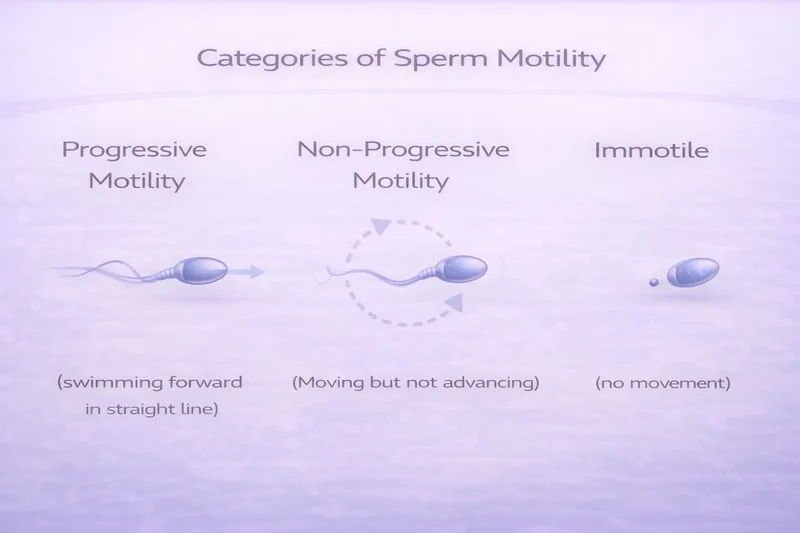
Your semen analysis report classifies sperm movement into three categories:
Progressive motility (PR) — Sperm actively swimming forward in a straight line or large circles. These are the swimmers that can actually reach the egg. The WHO 2021 reference is ≥30%.
Non-progressive motility (NP) — Sperm that are moving but not going anywhere useful—swimming in tight circles, vibrating on the spot, or flagellating without forward progress.
Immotile (IM) — Sperm showing no movement at all. They may be dead or alive but unable to move.
Total motility = PR + NP. The WHO 2021 reference is ≥42%.
Progressive motility is the measure that matters most for natural conception. A man with 50% total motility but only 10% progressive motility will likely have more difficulty conceiving than someone with 42% total motility but 35% progressive.
When Is Motility Considered "Low"?
Asthenozoospermia is diagnosed when either:
❌ Total motility is below 42%, OR ❌ Progressive motility is below 30%
But context matters enormously. A single result of 28% progressive motility doesn't necessarily mean you have a persistent problem. Semen parameters fluctuate naturally by 28–43% between consecutive samples due to factors like recent illness, stress, sleep quality, and even the time of year. That's why at least two analyses, taken 2–4 weeks apart, are recommended before making any clinical decisions.
It also matters whether low motility appears in isolation or alongside other abnormalities. Isolated asthenozoospermia—low motility with normal concentration and morphology—occurs in roughly 19% of infertile men and often has different underlying causes than combined abnormalities like OAT (oligoasthenoteratozoospermia).
Severity Grading
There's no formal WHO classification of asthenozoospermia severity, but clinicians generally consider:
Mild — Progressive motility 20–29% (just below reference)
Moderate — Progressive motility 10–19%
Severe — Progressive motility <10%, or complete asthenozoospermia (0% motile sperm, which is very rare and warrants urgent investigation for structural flagellar defects)
What Causes Low Sperm Motility?
The causes of asthenozoospermia range from easily correctable lifestyle factors to medical conditions requiring specialist treatment. In many cases, the cause remains unexplained even after thorough investigation—this is termed idiopathic asthenozoospermia. Here's what the evidence says about each known cause.

Medical Causes
Varicocele
A varicocele—an abnormal dilation of veins in the spermatic cord—is the most common identifiable and treatable cause of male infertility, found in approximately 15% of all men and up to 40% of infertile men. Varicoceles impair sperm motility through several mechanisms: increased scrotal temperature, elevated oxidative stress, accumulation of toxic metabolites, and disrupted blood flow to the testes.
The good news: varicocele repair (varicocelectomy) has strong evidence for improving motility. A large meta-analysis of 351 studies found that varicocelectomy significantly improved progressive sperm motility, with improvements typically visible within 3 months of surgery. Another meta-analysis found that total and progressive motility improved by approximately 10.9% and 9.7% respectively after repair.
Varicoceles are usually detected by physical examination—your GP or urologist can feel for enlarged veins above the testicle while you stand. Ultrasound confirms the diagnosis and grades severity.
Genital Tract Infections and Inflammation
Infections of the reproductive tract—including prostatitis, epididymitis, and sexually transmitted infections like chlamydia and gonorrhoea—can directly impair sperm motility. White blood cells recruited to fight infection produce reactive oxygen species that damage sperm membranes and mitochondria.
If your semen analysis shows leukocytospermia (>1 million white blood cells/mL), infection should be investigated with semen culture and, if indicated, treated with appropriate antibiotics. Motility often improves once the infection resolves.
Hormonal Imbalances
Sperm production and maturation depend on a finely tuned hormonal axis. Low testosterone (hypogonadism), elevated prolactin, or thyroid dysfunction can all impair sperm motility. Obesity-related hormonal disruption is particularly common—excess body fat increases aromatase activity, converting testosterone to oestradiol, which suppresses the hypothalamic-pituitary-gonadal axis and impairs spermatogenesis.
Hormonal testing (testosterone, FSH, LH, prolactin, thyroid function) should be part of the workup for unexplained asthenozoospermia.
Anti-Sperm Antibodies
Anti-sperm antibodies (ASAs) can bind to the sperm surface and directly impair motility by creating a drag effect or causing sperm to agglutinate (stick together). ASAs may develop after vasectomy reversal, testicular trauma, surgery, or infection. If your semen analysis shows agglutination (sperm clumping together), ASA testing should be considered.
Structural and Genetic Causes
In a minority of cases, low motility results from structural abnormalities of the sperm tail (flagellum). The flagellum is an extraordinarily complex structure containing over 1,000 different proteins arranged in a precise 9+2 microtubular architecture that generates the wave-like beating motion needed for propulsion.
Genetic mutations affecting flagellar structure can cause a range of motility defects:
Primary ciliary dyskinesia (PCD) — A genetic condition affecting both sperm flagella and cilia throughout the body. Men with PCD typically have nearly 100% immotile sperm alongside chronic sinusitis, bronchiectasis, and sometimes situs inversus (reversed organ positioning). If you have both severe asthenozoospermia and chronic respiratory symptoms, PCD should be investigated.
Multiple morphological abnormalities of the flagella (MMAF) — A group of genetic conditions causing visible flagellar defects (short, coiled, absent, or irregular tails) alongside severely impaired motility. Over 40 genes have now been linked to MMAF.
These structural causes are rare but important to identify because they have specific implications for fertility treatment—particularly whether ICSI is likely to succeed.
Lifestyle and Environmental Causes
This is where you have the most control. Lifestyle factors are a significant cause of impaired sperm quality in the modern world, and the good news is that changes here can lead to measurable improvements within one spermatogenesis cycle (approximately 74 days, or roughly 3 months).
Smoking
Smoking is one of the most well-documented causes of reduced sperm motility. A recent review found that smoking increases sperm DNA fragmentation by approximately 10% and alters hormonal profiles. The mechanism involves direct oxidative damage from tobacco's 4,000+ chemical constituents, including nicotine, cadmium, and lead, which generate reactive oxygen species that damage sperm membranes and mitochondria.
E-cigarettes may carry similar risks, though long-term data is still emerging.
Evidence for quitting: Sperm parameters typically begin improving within 3 months of smoking cessation, with full recovery taking up to 6 months (one complete spermatogenesis cycle).
Alcohol
Chronic alcohol consumption disrupts the hypothalamic-pituitary-gonadal axis, raises sperm DNA fragmentation, and can cause testicular atrophy. Heavy drinking is clearly harmful, but even moderate consumption may have effects—studies show combined tobacco and alcohol use has cumulative detrimental effects on progressive motility.
Practical advice: Aim for no more than 14 units per week (UK guidelines), spread across several days. If you're actively trying to conceive, reducing further or abstaining temporarily may help.
Obesity and Poor Diet
Excess body weight impairs sperm motility through multiple pathways: hormonal disruption (elevated oestradiol, reduced testosterone), increased scrotal temperature from adipose tissue, chronic inflammation, and oxidative stress. Men with a BMI over 25 are significantly more likely to have reduced motility and total sperm count.
The encouraging evidence: even modest weight loss improves sperm parameters. A 14-week weight loss programme was shown to increase total motile sperm count and reduce DNA fragmentation from 20.2% to 17.5%.
A Mediterranean-style diet—rich in fruits, vegetables, whole grains, fish, nuts, and olive oil—has been consistently associated with better sperm quality in observational studies.
Heat Exposure
The testes hang outside the body for a reason—sperm production requires a temperature 2–4°C below core body temperature. Anything that raises scrotal temperature can impair motility and overall sperm production.
Common heat sources affecting fertility:
❌ Hot baths, saunas, and hot tubs (regular use)
❌ Tight underwear
❌ Prolonged laptop use on the lap
❌ Extended sitting (long-haul driving, desk work)
❌ Occupational heat exposure (kitchens, bakeries, welding, furnaces)
The good news: heat-related damage is usually reversible once the source is removed. Switch to looser underwear, take breaks from sitting, and avoid direct heat sources near the groin.
Mobile Phone Radiation
There is growing evidence that carrying a mobile phone in your trouser pocket may affect sperm quality. A meta-analysis reported a mean reduction of 8.1% in motility associated with mobile phone exposure, likely through both thermal effects and RF-EMF-induced oxidative stress. While the evidence isn't conclusive enough to cause alarm, carrying your phone in a jacket or bag pocket rather than your trousers is a sensible low-cost precaution.
Recreational Drugs and Anabolic Steroids
Cannabis use has been shown to affect both spermatogenesis and sperm motility. Anabolic steroids—including testosterone replacement therapy—are particularly harmful, as exogenous testosterone shuts down the body's natural sperm production via negative feedback on FSH and LH. This can cause severe oligozoospermia or even azoospermia, and recovery after stopping may take months to over a year.
If you're using testosterone or anabolic steroids and want to conceive, speak to a specialist urgently—there are alternative approaches that support both testosterone levels and sperm production.
Medications
Several prescription medications can impair sperm motility. These include certain SSRIs (antidepressants), 5-alpha reductase inhibitors (finasteride/dutasteride for hair loss or prostate), some blood pressure medications, and chemotherapy agents. Always tell your fertility specialist about every medication you take, including over-the-counter and herbal supplements.
Environmental and Occupational Exposures
Endocrine-disrupting chemicals (EDCs) in the environment are increasingly linked to declining sperm quality. Pesticides, plasticisers (BPA, phthalates), heavy metals, and industrial chemicals can all impair sperm function through oxidative stress and hormonal disruption. While individual exposure is hard to control completely, practical steps include avoiding plastic food containers (especially when heated), choosing organic produce where possible, and minimising occupational chemical exposure with appropriate protective equipment.
What Actually Helps: Evidence-Based Treatments
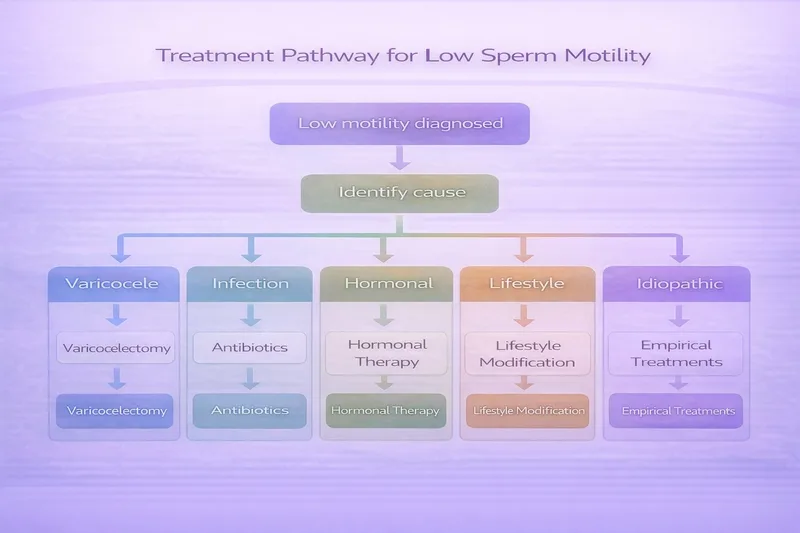
Step 1: Identify and Treat the Underlying Cause
The most effective approach to improving sperm motility is to find and address the root cause. This is why a proper clinical workup matters—it's not just about supplements and lifestyle tips.
If varicocele is present: Microsurgical varicocelectomy has the strongest evidence, with significant improvements in motility seen within 3 months. The microsurgical subinguinal approach has the lowest recurrence rate and fewest complications.
If infection is present: Appropriate antibiotic treatment for documented genital tract infections can improve motility once the infection and inflammation resolve.
If hormonal imbalance is present: Depending on the specific issue, treatment may include clomiphene citrate (to stimulate FSH/LH), hCG injections, or management of elevated prolactin or thyroid dysfunction. Weight loss in obese men can itself normalise hormonal profiles.
Step 2: Lifestyle Modifications
Regardless of the underlying cause, lifestyle changes should be implemented in parallel with medical treatment. These have solid evidence behind them and no downside:
✅ Stop smoking — Improvements within 3 months
✅ Reduce or eliminate alcohol — Especially if heavy use
✅ Achieve a healthy weight — Even modest loss (5–10%) helps
✅ Eat a Mediterranean-style diet — Rich in antioxidants, omega-3s, zinc, selenium
✅ Exercise regularly — Moderate exercise improves sperm quality; avoid excessive endurance training which may temporarily reduce parameters
✅ Avoid heat exposure — Loose boxers, no hot tubs, laptop on desk not lap
✅ Manage stress — Chronic stress hormones can interfere with testosterone production
✅ Sleep 7–8 hours — Poor sleep quality and short sleep duration are associated with reduced sperm motility
✅ Move your phone — Out of trouser pockets
Allow at least 3 months for lifestyle changes to show results, as this represents a full spermatogenesis cycle.
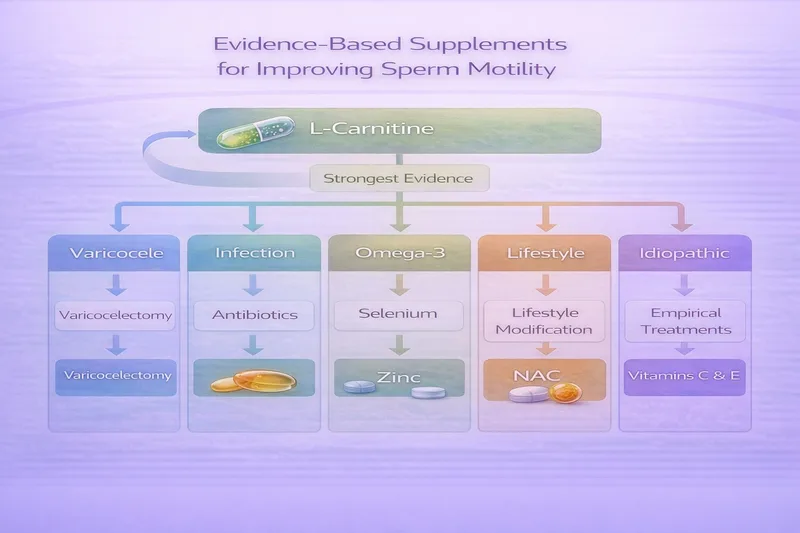
Step 3: Targeted Supplementation
Antioxidant supplementation is widely used for male infertility, and the evidence—while mixed—is generally positive for motility improvement specifically. A meta-analysis of 45 RCTs (4,332 patients) found that antioxidant therapy significantly improved sperm concentration, motility, and morphology compared to placebo or no treatment, and increased spontaneous pregnancy rates (OR 1.97).
However, it's important to note that a large multicentre placebo-controlled trial found no benefit after 3 months of combination antioxidant therapy, though this study was criticised for methodological limitations.
Here's what the evidence says about specific supplements for motility:
Best Evidence for Motility
L-Carnitine (2g/day) + L-Acetyl Carnitine (1g/day) — A network meta-analysis ranked carnitine as the most effective antioxidant for improving sperm motility (SUCRA: 88.7%), with a mean improvement of +12.43% versus placebo. Carnitine is essential for mitochondrial energy production in the sperm midpiece—the engine that powers flagellar movement. Multiple RCTs have confirmed significant motility improvements, particularly in men with lower baseline motility.
CoQ10 (200–400mg/day) — Ranked second for motility improvement in the same network meta-analysis, with a mean improvement of +7.33% versus placebo. CoQ10 is a key component of the mitochondrial electron transport chain and a potent antioxidant. It also showed the strongest evidence for improving sperm concentration.
Omega-3 Fatty Acids (DHA + EPA, ~1,840mg/day) — Shown to improve motility, concentration, and morphology after 32 weeks of supplementation. DHA is a structural component of the sperm membrane, and adequate levels are important for membrane fluidity and function.
Supporting Evidence
Selenium (200μg/day) — An essential trace element for selenoprotein production, which protects sperm from oxidative damage. Network meta-analysis showed it was more effective than placebo for motility improvement.
Zinc (30mg/day) — Important for testosterone synthesis and sperm membrane stability. Deficiency is associated with reduced motility.
N-Acetyl Cysteine / NAC (600mg/day) — A precursor to glutathione, the body's master antioxidant. A randomised trial showed significant reduction in DNA fragmentation and improved sperm parameters after 3 months.
Vitamin C (500–1,000mg/day) + Vitamin E (400 IU/day) — Evidence for these individually is modest, but combination use may reduce DNA damage.
For detailed dosing, evidence grades, and UK product recommendations, see: Male Fertility Supplements UK: Evidence-Based Guide 2026
Important caveats about supplementation:
⚠️ Supplements work best when combined with lifestyle changes—they're not a substitute
⚠️ Allow at least 3 months before expecting results (a full spermatogenesis cycle)
⚠️ More is not necessarily better—excessive antioxidant intake can cause "reductive stress" which may itself harm sperm
⚠️ Supplements should complement, not replace, medical investigation and treatment
⚠️ Discuss any supplementation with your GP or fertility specialist
When Motility Alone Isn't Enough: Assisted Reproduction
If motility remains low despite treatment, or if time is a factor (particularly if your partner is over 35), assisted reproduction may be recommended.
IUI (Intrauterine Insemination)
Sperm washing and concentration followed by direct placement into the uterus can bypass the cervical barrier, which is one of the main obstacles for poorly motile sperm. IUI is typically considered when:
✅ Progressive motility is mildly reduced but not severely impaired ✅ Total motile sperm count after washing is >5 million ✅ Female partner has patent fallopian tubes and regular ovulation ✅ There are no other significant male or female factors
IVF (In Vitro Fertilisation)
Standard IVF places sperm and eggs together in a dish, requiring sperm to fertilise the egg independently. This requires reasonable motility—it's less suitable for severe asthenozoospermia.
ICSI (Intracytoplasmic Sperm Injection)
ICSI involves injecting a single sperm directly into the egg, bypassing the need for motility entirely. It's the treatment of choice when:
❌ Severe asthenozoospermia is present
❌ Very few motile sperm are available
❌ Previous IVF has failed
❌ Structural flagellar defects prevent natural movement
Even with ICSI, the selection of morphologically normal, vital sperm is important—immotile but viable sperm can still achieve fertilisation through ICSI if they are alive (assessed by hypo-osmotic swelling test or laser-assisted selection).
Advanced Sperm Selection Techniques
Newer techniques including IMSI (intracytoplasmic morphologically selected sperm injection), MACS (magnetic-activated cell sorting), and microfluidic sperm sorting are being developed to select the healthiest sperm for ICSI, even from severely compromised samples.
Monitoring Progress: What to Expect
Timeline for Improvement
Because spermatogenesis takes approximately 74 days, any intervention—whether lifestyle change, supplementation, surgery, or medical treatment—needs at least 3 months before results are visible on a repeat semen analysis.
After varicocelectomy: Maximum improvement typically occurs within 3 months, with no further significant improvement beyond that point.
After lifestyle changes: Gradual improvement over 3–6 months.
After starting supplements: Allow 3 months minimum; some studies use 6-month protocols.
After stopping harmful medications/substances: Recovery time varies—3 months for smoking, potentially 6–12+ months for anabolic steroids.
Follow-Up Testing
Request a repeat semen analysis 3 months after making changes or starting treatment. If motility has improved but you're still not conceiving, consider:
DNA fragmentation testing — Normal motility doesn't exclude DNA damage Hormonal reassessment — Particularly if motility hasn't improved as expected Partner evaluation — Female factors must be investigated in parallel
Frequently Asked Questions
Can low motility fix itself?
Sometimes, yes. If the cause is temporary—a recent fever, acute illness, stress, medication, or lifestyle factor—motility may normalise on its own once the trigger resolves. This is why a repeat analysis is always recommended before making clinical decisions. However, if motility is consistently low across two or more tests, investigation and treatment are warranted.
My concentration and morphology are normal but motility is low—why?
Isolated asthenozoospermia affects roughly 19% of infertile men. It suggests the issue is specifically with sperm function or the flagellar machinery rather than overall sperm production. Causes include mitochondrial dysfunction, structural flagellar defects, anti-sperm antibodies, genital tract infection/inflammation, or oxidative stress. It's also particularly responsive to antioxidant supplementation since the mitochondria powering the flagellum are especially vulnerable to oxidative damage.
Is 20% progressive motility enough to conceive naturally?
It's possible, though the probability is reduced compared to normal motility. Fertility is a spectrum, not a binary. Men with moderately reduced motility do conceive naturally—it may just take longer. Factors like your partner's age and fertility, timing of intercourse, and other semen parameters all play a role. If you've been trying for over 12 months (6 months if your partner is over 35), seek specialist advice rather than waiting.
Does abstinence period affect motility?
Yes. Longer abstinence periods (>5 days) are associated with decreased motility and increased DNA fragmentation due to sperm accumulating reactive oxygen species during storage in the epididymis. Conversely, very short abstinence (<1 day) may reduce volume and total count. The optimal window is 2–5 days. Some emerging evidence suggests shorter abstinence periods may actually improve motility parameters, though this is still debated.
Should I take testosterone to improve my sperm?
Absolutely not without specialist guidance. Exogenous testosterone (TRT, steroids) paradoxically shuts down the body's natural sperm production by suppressing FSH and LH through negative feedback. This can cause severe oligozoospermia or azoospermia. If you have low testosterone and want to conceive, a fertility specialist can prescribe alternatives like clomiphene citrate or hCG that raise testosterone while maintaining or improving sperm production.
How quickly do supplements start working?
You won't see changes overnight. Because spermatogenesis takes approximately 74 days, you need at least one full cycle (3 months) of consistent supplementation before retesting. Some studies showing benefit used 6-month protocols. Start supplements early and be consistent—don't judge effectiveness before the 3-month mark.
Can stress really affect motility?
Yes. Chronic psychological stress elevates cortisol, which can suppress testosterone production and impair spermatogenesis. Additionally, stress often leads to secondary factors that further impair motility—poor sleep, increased alcohol intake, unhealthy eating, and reduced exercise. Addressing stress through your preferred methods (exercise, mindfulness, therapy, work-life balance) is a legitimate part of fertility optimisation.
My GP says my motility is "borderline"—should I worry?
A borderline result (e.g., progressive motility of 25–29%) on a single test is genuinely common and may reflect normal variation rather than a persistent problem. Request a repeat test in 2–4 weeks. In the meantime, implementing the lifestyle changes described above carries no risk and may improve your result. If the second test confirms borderline or low motility, discuss further investigation with your GP.
When to Seek Specialist Help
Don't delay seeking a referral to a urologist or fertility specialist if:
❌ Two semen analyses show progressive motility below 30%
❌ You've been trying to conceive for 12 months without success (6 months if your partner is over 35)
❌ You have complete asthenozoospermia (0% motile sperm) on any test
❌ You have a known or suspected varicocele
❌ You have chronic respiratory symptoms alongside severe motility issues (suggesting possible PCD)
❌ You're using or have recently stopped anabolic steroids or testosterone
❌ Motility hasn't improved after 3–6 months of lifestyle changes and supplementation
Low sperm motility is common, often improvable, and rarely the end of the road. Whether through lifestyle changes, targeted supplementation, medical treatment, or assisted reproduction, there are evidence-based options at every stage. The most important step is getting proper investigation—and starting sooner rather than later.
Related Articles
Understanding Semen Analysis Results: Complete UK Guide
Male Fertility Supplements UK: Evidence-Based Guide 2026
Prenatal Vitamins UK: What You Actually Need When Trying to Conceive
When to See a Fertility Doctor UK: Guide for Trying to Conceive
Medical Disclaimer This article is for informational and educational purposes only and is not intended as medical advice. Always consult with your GP or a qualified fertility specialist for personalised guidance regarding sperm motility or any fertility concerns. The information provided should not replace professional medical consultation or delay seeking appropriate care. If you have been trying to conceive for over 12 months (or 6 months if your partner is over 35), seek professional evaluation without delay.