If you've been referred for a semen analysis—or you're considering one—the results can feel overwhelming. Pages of numbers, unfamiliar medical terms, and percentages that seem to mean everything and nothing at once. Here's something reassuring: understanding your semen analysis is far more straightforward than it first appears, and it's one of the most important steps you can take on your fertility journey.
Semen analysis is the cornerstone of male fertility evaluation. It's typically the first investigation your GP or fertility specialist will request, and for good reason: male factor infertility contributes to approximately 50% of all infertility cases. Yet despite its importance, many men receive their results without adequate explanation of what the numbers actually mean—or what to do next.
This comprehensive guide walks you through every parameter on a semen analysis report, explains what the World Health Organization (WHO) 2021 reference values mean in practice, covers the advanced tests your standard analysis doesn't include, and helps you understand when further investigation is warranted.
Also see: Male Fertility Supplements UK: Evidence-Based Guide 2026 for improving sperm parameters through targeted supplementation.

What Is a Semen Analysis?
A semen analysis—sometimes called a seminogram or sperm test—is a laboratory examination of your ejaculate that evaluates both the fluid (semen) and the sperm cells it contains. The test measures multiple parameters across two categories:
Macroscopic examination (what the laboratory can assess with the naked eye): volume, appearance, liquefaction time, viscosity, and pH.
Microscopic examination (what requires a microscope): sperm concentration, total count, motility, morphology, vitality, and the presence of other cells.
Together, these measurements provide a detailed snapshot of your reproductive health and help identify potential issues affecting fertility. However, it's important to understand that semen analysis is a screening tool—not a definitive fertility diagnosis. The WHO 6th Edition Manual emphasises that reference values alone are insufficient to diagnose male infertility, and further clinical evaluation is needed. A single result doesn't tell the whole story, which is why at least two analyses are usually recommended.

Getting a Semen Analysis in the UK
The NHS Route
To access a semen analysis through the NHS, you'll typically need a GP referral. Most GPs will refer couples who have been trying to conceive for 12 months without success (or 6 months if the female partner is over 35). Your GP arranges the referral to a hospital andrology laboratory, where you'll be given an appointment.
The NHS waiting time varies by area. Some hospitals offer appointments within a few weeks; others may have waits of 10–12 weeks. Results are usually sent to your referring GP or consultant within 5–10 working days.
NHS semen analysis is free at the point of care, but the referral criteria and waiting times mean it's not always ideal for men who want proactive early testing.
Private Testing
Private semen analysis is available throughout the UK at fertility clinics and some independent laboratories. Costs typically range from £100–£250 for a standard analysis. Some private services offer home collection kits, though producing the sample on-site is generally preferred for accuracy—the sample should be analysed within 60 minutes of production.
Private testing can be accessed without a GP referral and usually has much shorter waiting times, making it a good option for men who want to check their fertility status early.
At-Home Testing Kits
Several at-home sperm testing kits are now available in the UK. These typically measure sperm concentration and sometimes motility, but they do not provide the comprehensive analysis available in a clinical laboratory. They can be useful as an initial screen but should not replace a full laboratory semen analysis if you have concerns about fertility.

How to Prepare for Your Semen Analysis
Preparation matters—the quality of your sample can significantly affect your results. Follow these guidelines to ensure the most accurate analysis.
Abstinence Period
The WHO recommends 2–7 days of sexual abstinence (no ejaculation) before producing your sample. Most UK clinics request 2–5 days as the optimal window.
Why this matters: Too short an abstinence period (less than 2 days) may result in lower volume and concentration, though recent research suggests shorter abstinence may actually improve motility and reduce DNA fragmentation. Too long (more than 7 days) can decrease motility and increase the proportion of dead or damaged sperm, as sperm accumulate reactive oxygen species during extended storage in the epididymis.
If you're having a repeat analysis, try to maintain the same abstinence period as your first test for meaningful comparison.
Sample Collection
Producing the sample at the clinic is the gold standard. Most andrology departments provide private rooms for this purpose. The sample should be collected by masturbation into a sterile, non-toxic container provided by the laboratory.
Important collection guidelines:
✅ Wash and dry your hands and penis thoroughly before collection
✅ Collect the entire ejaculate—the first fraction contains the highest sperm concentration
✅ Use only the sterile container provided
❌ Do not use lubricants, saliva, or condoms (unless specifically designed for semen collection)—these can be toxic to sperm
❌ Do not collect via interrupted intercourse (coitus interruptus), as you may lose the sperm-rich first portion
If you must produce the sample at home, keep it at body temperature (inside your clothing, close to your body) and deliver it to the laboratory within 30–60 minutes of collection.
Other Preparation Tips
Avoid excessive alcohol for 2–3 days before the test
Inform the laboratory of any recent illness, fever, or medications—illness in the previous 3 months can temporarily affect sperm production
Don't skip the test if you're feeling unwell on the day—but do inform the laboratory so they can note it on your report
Avoid hot baths, saunas, or strenuous exercise for 2–3 days before collection

Understanding Your Results: WHO 2021 Reference Values
Your semen analysis report will contain multiple parameters, each compared against reference values established by the World Health Organization. The most recent reference values come from the WHO 6th Edition Manual (2021), which updated the previous 2010 criteria.
What Do "Reference Values" Actually Mean?
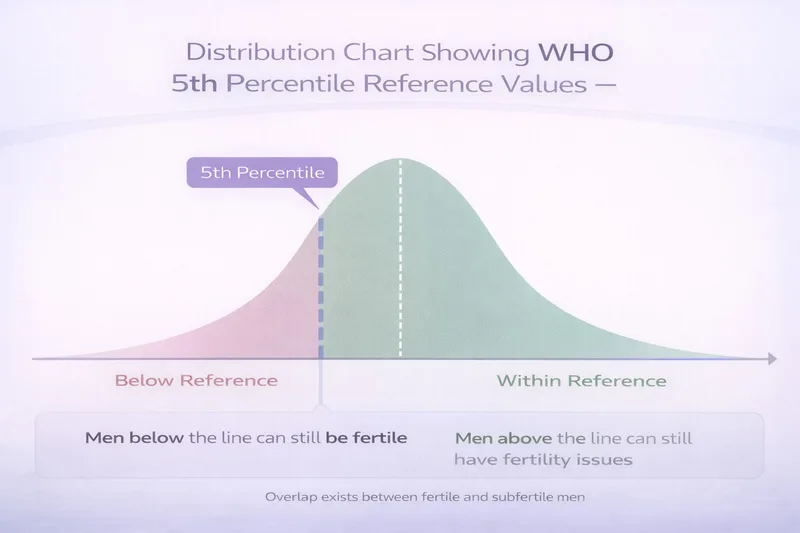
This is crucial to understand: WHO reference values are not pass/fail thresholds. They represent the 5th percentile of semen parameters from men whose partners conceived within 12 months of trying. This means:
Being above the reference value doesn't guarantee fertility—some men with "normal" results still struggle to conceive.
Being below the reference value doesn't mean you're infertile—many men with below-reference results father children naturally.
The 6th edition emphasises that these values are a starting point for clinical assessment, not a definitive diagnosis. Your fertility specialist will interpret your results in the context of your overall health, your partner's fertility, and your reproductive history.
Parameter-by-Parameter Breakdown
Here's what each measurement on your report means:
Semen Volume
📊 WHO 2021 reference: ≥1.4 mL
Semen volume measures the total amount of ejaculate produced. The fluid comes primarily from the seminal vesicles (65–75%) and the prostate gland (25–30%), with smaller contributions from the bulbourethral glands and epididymis.
What your result means:
≥1.4 mL → Normal Adequate volume within the reference range.
<1.4 mL → Hypospermia May indicate incomplete collection, short abstinence, ejaculatory duct obstruction, retrograde ejaculation, or seminal vesicle dysfunction.
>6 mL → Hyperspermia Usually not clinically significant; may dilute sperm concentration.
0 mL → Aspermia No ejaculate produced—may indicate retrograde ejaculation or ejaculatory duct obstruction.
Low volume is one of the parameters most affected by collection issues, so if your volume is low, your clinician may ask whether the entire sample was collected.
Liquefaction Time
📊 WHO 2021 reference: Complete within 60 minutes (typically 15–30 minutes)
Immediately after ejaculation, semen forms a coagulum (gel-like consistency). This is normal—it's caused by proteins from the seminal vesicles. Over the next 15–30 minutes, enzymes from the prostate (primarily prostate-specific antigen, or PSA) break down this gel, allowing the semen to become fluid. This process is called liquefaction.
What your result means:
Normal liquefaction: Semen becomes completely fluid within 15–30 minutes. This is reported as "complete."
Delayed or incomplete liquefaction: If the sample hasn't liquefied within 60 minutes, it may indicate prostate dysfunction or infection. However, it can also be an artefact of collection or temperature. Delayed liquefaction can trap sperm and reduce motility assessment accuracy.
Semen pH
📊 WHO 2021 reference: ≥7.2
The pH of semen reflects the balance of acidic prostatic secretions and alkaline seminal vesicle fluid. Normal semen is slightly alkaline.
What your result means:
≥7.2 → Normal range
<7.2 (acidic) → May indicate seminal vesicle dysfunction, ejaculatory duct obstruction, or contamination with urine. If combined with low volume and absence of sperm (azoospermia), it may suggest congenital bilateral absence of the vas deferens (CBAVD)—a condition linked to cystic fibrosis carrier status.
pH alone is rarely diagnostic, but an abnormally low pH combined with low volume and azoospermia is a clinically important pattern that warrants further investigation.
Sperm Concentration
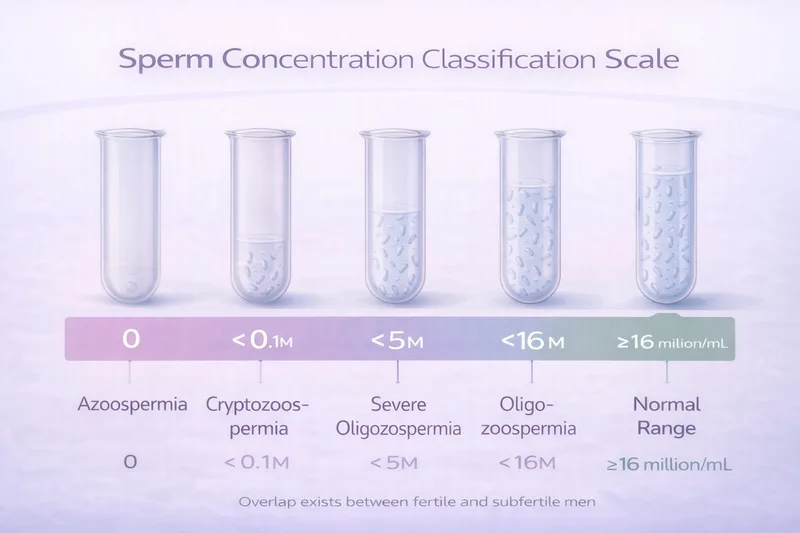
📊 WHO 2021 reference: ≥16 million per millilitre (million/mL)
Sperm concentration—sometimes called sperm density—measures how many sperm cells are present per millilitre of semen. This is one of the most commonly discussed parameters and reflects testicular sperm production capacity.
What your result means:
≥16 million/mL → Normal concentration Within the reference range.
<16 million/mL → Oligozoospermia Reduced concentration—may impact fertility.
<5 million/mL → Severe oligozoospermia Significantly reduced—genetic testing may be recommended.
<0.1 million/mL → Cryptozoospermia Very few sperm found only after centrifuging the sample.
0 → Azoospermia No sperm detected—requires further investigation (obstructive vs non-obstructive).
Oligozoospermia has many potential causes, including hormonal imbalances, varicocele, genetic factors, lifestyle factors, and environmental exposures. Azoospermia requires specialist investigation to determine whether it's caused by a blockage (obstructive) or a production problem (non-obstructive), as the treatment pathways are very different.
Total Sperm Count
📊 WHO 2021 reference: ≥39 million per ejaculate
Total sperm count is calculated by multiplying sperm concentration by semen volume. It represents the total number of sperm in your entire ejaculate and is considered a strong predictor of fertility potential because it accounts for both sperm density and fluid volume.
For example: A man with 20 million/mL concentration and 3 mL volume has a total count of 60 million (normal). A man with 20 million/mL but only 1 mL volume has a total count of 20 million (below reference).
Sperm Motility
📊 WHO 2021 reference: ≥42% total motility, ≥30% progressive motility
Motility—the ability of sperm to move—is arguably the most important parameter for natural conception, because sperm must actively swim through the cervix, uterus, and fallopian tubes to reach the egg.
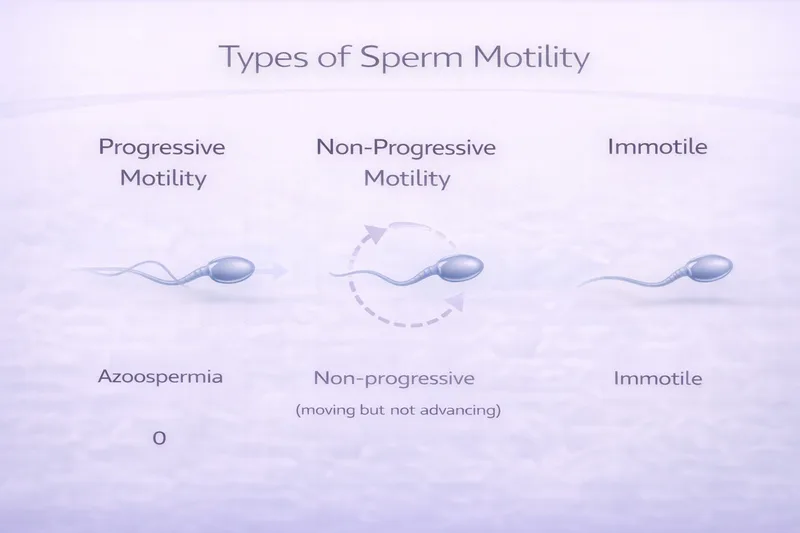
Your report will classify sperm movement into three categories:
Progressive motility (PR): Sperm moving actively forward in a straight line or large circles. These are the sperm most likely to reach and fertilise an egg. Reference: ≥30%.
Non-progressive motility (NP): Sperm moving but not making forward progress—swimming in tight circles, flagellating, or vibrating in place. These sperm are alive but unlikely to reach the egg naturally.
Immotile (IM): Sperm showing no movement at all. These may be dead or alive but non-functional.
Total motility = PR + NP. Reference: ≥42%.
What your result means:
≥42% total, ≥30% progressive → Normal motility Within the reference range.
<42% total or <30% progressive → Asthenozoospermia Reduced motility—may be caused by varicocele, infection, antisperm antibodies, oxidative stress, or lifestyle factors.
If motility is very low, your clinician may recommend a vitality test to determine whether immotile sperm are dead (necrozoospermia) or alive but unable to move (which may suggest structural defects such as primary ciliary dyskinesia).
Sperm Morphology
📊 WHO 2021 reference: ≥4% normal forms (strict Kruger criteria)
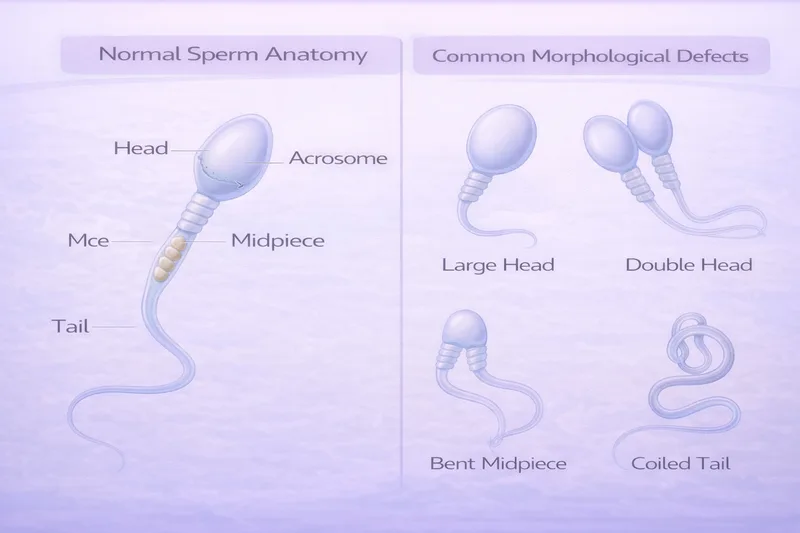
Morphology assesses the shape and structure of sperm. Each sperm is evaluated for defects in three regions:
Head: Should be smooth, oval-shaped, with a well-defined acrosome (cap) covering 40–70% of the head. Defects include large heads, small heads, tapered heads, round heads, double heads, or vacuoles.
Midpiece: Should be slender, straight, and approximately 1.5× the head length. It contains the mitochondria that power sperm movement. Defects include thick, irregular, or bent midpieces, and residual cytoplasmic droplets.
Tail: Should be thinner than the midpiece, uncoiled, and approximately 45 μm long. Defects include short, multiple, coiled, or broken tails.
A sperm must have a perfectly normal head, midpiece, AND tail to be classified as morphologically normal. This is why even fertile men typically have low morphology scores—the criteria are extremely strict.
What your result means:
≥4% normal → Normal morphology Within the reference range.
<4% normal → Teratozoospermia Low percentage of normally shaped sperm—common and doesn't necessarily prevent conception.
0% normal → Severe teratozoospermia No normally shaped sperm—may warrant further investigation, though pregnancy is still possible with assisted reproduction.
Morphology is one of the most subjective and variable parameters in semen analysis. Different technicians and laboratories can score the same sample differently, which is why morphology results should always be interpreted alongside other parameters rather than in isolation.
Sperm Vitality
📊 WHO 2021 reference: ≥54% live sperm
Vitality testing determines what proportion of sperm are alive versus dead. It's typically performed when motility is low (particularly when <40% of sperm are motile), to distinguish between dead sperm and live-but-immotile sperm.
The most common method is the eosin-nigrosin stain: dead sperm have damaged membranes that allow dye to enter, staining them pink/red, while live sperm with intact membranes remain unstained (white).
What your result means:
≥54% live → Normal vitality Within the reference range.
<54% live → Necrozoospermia High proportion of dead sperm—may indicate epididymal dysfunction, prolonged abstinence, or toxin exposure.
If a large proportion of sperm are alive but immotile, this suggests a structural problem with the sperm tail (flagellum) and warrants investigation for conditions such as primary ciliary dyskinesia or ultrastructural flagellar defects.
Additional Parameters
Your report may also include:
Appearance/Colour: Normal semen is grey-opalescent. A yellow tinge may indicate prolonged abstinence, jaundice, or certain medications. A reddish-brown colour (haemospermia) suggests blood in the semen, which may be caused by infection, inflammation, or (rarely) more serious conditions and should be investigated.
Viscosity: Normal semen leaves the pipette in small, discrete drops. Highly viscous semen forms long strands and may interfere with sperm motility. High viscosity can be caused by infection, inflammation, or dehydration.
Round cells: The presence of round cells in semen may represent immature germ cells (shed from the seminiferous tubules) or white blood cells (leukocytes). If round cell concentration is high, peroxidase staining can differentiate between the two.
Leukocytes (white blood cells): A concentration >1 million/mL is termed leukocytospermia and may indicate infection or inflammation in the reproductive tract. This warrants further investigation, potentially including semen culture, as genital tract infection can significantly impair sperm function.
Agglutination: Sperm sticking to each other (head-to-head, tail-to-tail, or mixed) suggests the possible presence of anti-sperm antibodies. This is different from aggregation (sperm sticking to debris or non-motile cells), which is usually not clinically significant.
Anti-sperm antibodies (ASAs): If agglutination is observed, testing for anti-sperm antibodies may be recommended. ASAs can interfere with sperm motility, cervical mucus penetration, and fertilisation. They may develop after vasectomy reversal, testicular injury, or infection.
Quick Reference: WHO 2021 Semen Analysis Reference Values
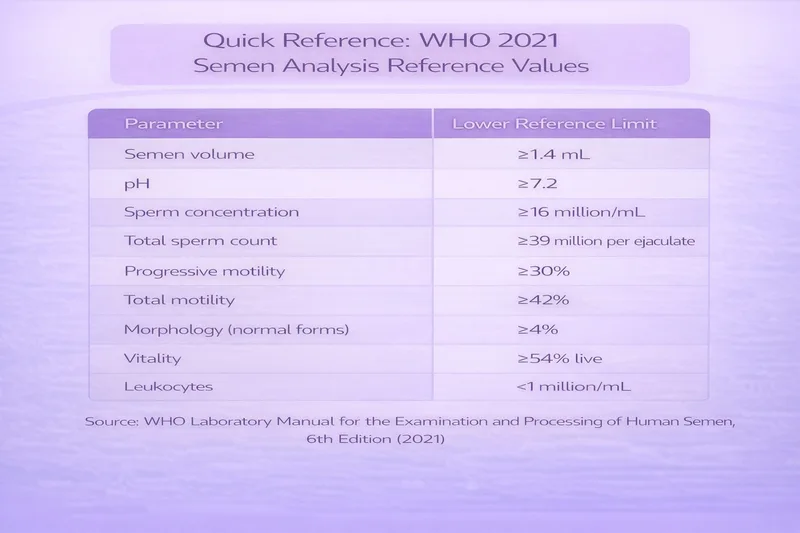
Semen volume: ≥1.4 mL pH: ≥7.2 Sperm concentration: ≥16 million/mL Total sperm count: ≥39 million per ejaculate Progressive motility: ≥30% Total motility: ≥42% Morphology (normal forms): ≥4% Vitality: ≥54% live Leukocytes: <1 million/mL
Source: WHO Laboratory Manual for the Examination and Processing of Human Semen, 6th Edition (2021)
Common Diagnostic Terms on Your Report
Your report may use specific medical terminology to describe patterns of abnormal results. Here's a quick-reference guide:
Normozoospermia — All measured parameters within WHO reference values
Oligozoospermia — Sperm concentration below reference (<16 million/mL)
Asthenozoospermia — Reduced sperm motility (<42% total or <30% progressive)
Teratozoospermia — Reduced normal morphology (<4% normal forms)
Oligoasthenozoospermia (OA) — Low concentration AND low motility
Oligoasthenoteratozoospermia (OAT) — Low concentration, motility, AND morphology—the most common combined abnormality
Azoospermia — No sperm detected in the ejaculate
Cryptozoospermia — Very few sperm found only after centrifugation
Necrozoospermia — High proportion of dead sperm
Leukocytospermia — Elevated white blood cells in semen (>1 million/mL)
Hypospermia — Low semen volume (<1.4 mL)
Haemospermia — Blood in the semen
Important: These terms describe patterns in your semen analysis—they are not diagnoses in themselves. The underlying cause of any abnormality requires further clinical investigation.
What Standard Semen Analysis Doesn't Tell You
A standard semen analysis is an excellent first-line screening tool, but it has significant limitations. Several important aspects of sperm function are not assessed in routine testing:
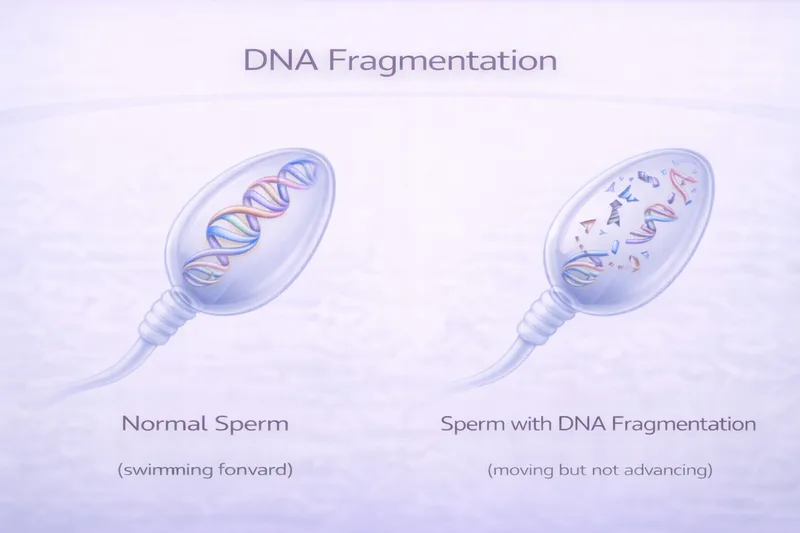
Sperm DNA Fragmentation (SDF)
Standard analysis examines sperm from the outside—their number, movement, and shape. It cannot assess the integrity of the DNA packaged inside each sperm head. Sperm DNA fragmentation refers to breaks or damage in the sperm's genetic material, which can impair fertilisation, embryo development, implantation, and pregnancy outcomes—even when conventional semen parameters appear completely normal.
SDF testing is particularly recommended when:
❌ Unexplained infertility (normal semen analysis, no female factor identified)
❌ Recurrent miscarriage (two or more pregnancy losses)
❌ Recurrent IVF/ICSI failure despite good embryo quality
❌ Varicocele (a common cause of elevated DNA fragmentation)
❌ Advanced paternal age (DNA fragmentation tends to increase with age)
❌ Exposure to known DNA-damaging factors (smoking, toxins, heat)
Testing Methods
Four main methods are used to measure sperm DNA fragmentation, each with different strengths:
TUNEL assay (Terminal deoxynucleotidyl transferase dUTP Nick End Labelling): Directly labels DNA strand breaks using fluorescent markers. Can be performed via flow cytometry (measuring thousands of sperm for statistical robustness) or light microscopy. Considered highly sensitive. Clinical threshold: approximately 20–22% DFI.
SCSA (Sperm Chromatin Structure Assay): Uses controlled acid denaturation and acridine orange staining to measure DNA susceptibility to damage. Performed exclusively by flow cytometry, giving it excellent standardisation and reproducibility. Also measures high DNA stainability (HDS), indicating immature sperm. Clinical threshold: approximately 20% DFI. The SCSA has the most extensive published clinical validation data.
SCD test (Sperm Chromatin Dispersion / Halosperm): Assesses whether sperm DNA forms a "halo" of dispersed loops after protein removal. Fragmented DNA fails to produce a halo. Performed by light microscopy, making it accessible to laboratories without flow cytometry equipment. Clinical threshold: approximately 25% DFI. Lower cost and equipment requirements, but analyses fewer sperm per sample.
Comet assay: Separates fragmented DNA through electrophoresis, creating a "comet tail" of migrating damaged DNA. The alkaline version detects both single- and double-strand breaks. Strong predictive power for infertility, but less standardised than SCSA or TUNEL.
Research shows these tests correlate well with each other, but they measure slightly different aspects of DNA damage. No single test has been established as the definitive gold standard, though SCSA and TUNEL (via flow cytometry) are the most widely used in clinical practice.
SDF testing is available privately in the UK from specialist fertility clinics and some NHS centres, typically costing £200–£400. Results are usually available within 1–2 weeks.
Also see: Male Fertility Supplements UK: Evidence-Based Guide 2026 for evidence on antioxidants that may help reduce DNA fragmentation, including NAC and CoQ10.

Oxidative Stress Measurement
Reactive oxygen species (ROS) testing measures the balance between free radical production and antioxidant capacity in semen. Elevated oxidative stress is implicated in 25–87% of male subfertility cases and damages sperm through lipid peroxidation of membranes, direct DNA strand breaks, and impaired mitochondrial function.
While not yet part of routine clinical practice, oxidative stress measurement can help identify men who may benefit from antioxidant supplementation. The WHO 6th Edition Manual includes oxidative stress testing as an "advanced examination."
Genetic Testing
Genetic abnormalities account for an estimated 15–30% of severe male infertility cases. Genetic testing may be recommended when:
Severe oligozoospermia (<5 million/mL) or azoospermia is identified
There is a family history of genetic conditions
Congenital bilateral absence of the vas deferens (CBAVD) is suspected
Common genetic tests include:
Karyotype analysis: Examines chromosome number and structure. Klinefelter syndrome (47,XXY) is the most common chromosomal cause of azoospermia or severe oligozoospermia.
Y-chromosome microdeletion testing: Deletions in the AZF (azoospermia factor) regions of the Y chromosome can cause impaired or absent sperm production. The type of deletion (AZFa, AZFb, or AZFc) has implications for whether sperm retrieval for IVF/ICSI is likely to be successful.
Cystic fibrosis (CFTR) gene testing: Mutations in the CFTR gene cause congenital bilateral absence of the vas deferens in approximately 2% of infertile men and up to 25% of men with obstructive azoospermia. Both partners should be tested, as carrier status has implications for offspring.
Hormonal Assessment
Hormone testing isn't part of semen analysis itself, but is frequently ordered alongside it when abnormalities are found. Key hormones include:
Testosterone: Low levels may indicate hypogonadism, affecting sperm production and sexual function.
FSH (Follicle-Stimulating Hormone): Elevated FSH suggests the testes are not producing sperm efficiently and the pituitary is working harder to compensate. Very high FSH with azoospermia suggests non-obstructive azoospermia (a production problem rather than a blockage).
LH (Luteinising Hormone): Works alongside FSH to stimulate testosterone production.
Prolactin: Elevated levels can suppress GnRH, reducing testosterone and sperm production.
Oestradiol: Elevated in obese men due to aromatase activity in fat tissue, potentially suppressing sperm production.
Interpreting Your Results: Putting It All Together

Scenario 1: All Parameters Normal (Normozoospermia)
A normal semen analysis is reassuring, but it doesn't guarantee fertility. Approximately 30% of men with normal semen parameters have abnormal sperm function that isn't detected by standard analysis. If you and your partner have been trying for over 12 months with normal results:
Consider sperm DNA fragmentation testing
Ensure your partner has had a comprehensive fertility evaluation
Review timing and frequency of intercourse around the fertile window
Discuss further investigation with a fertility specialist
Scenario 2: One Mildly Abnormal Parameter
A single mildly abnormal result on one analysis is very common and may not be clinically significant. Semen parameters fluctuate naturally—studies show variation of 28–43% between consecutive samples for concentration, motility, and morphology. Factors like recent illness, stress, poor sleep, alcohol, or even the time of year can temporarily affect results.
Your clinician will almost certainly recommend a repeat analysis 2–4 weeks (or up to 3 months) later. If the second sample is normal, the first result was likely a temporary fluctuation.
Scenario 3: Multiple Abnormal Parameters (e.g., OAT)
When concentration, motility, and morphology are all reduced (oligoasthenoteratozoospermia, or OAT), this suggests a more systemic issue with sperm production. Common causes include varicocele, hormonal imbalances, genetic factors, lifestyle factors, or oxidative stress.
Your GP should refer you to a urologist or andrologist for:
Physical examination (checking for varicocele, testicular size)
Hormone testing (testosterone, FSH, LH)
Further investigation based on severity
Lifestyle modification is recommended as first-line intervention alongside medical evaluation.
Also see: Male Fertility Supplements UK: Evidence-Based Guide 2026 for detailed evidence on CoQ10, L-carnitine, selenium, zinc, and other supplements that may improve sperm parameters.
Scenario 4: Azoospermia (No Sperm Found)
Azoospermia affects approximately 1% of all men and 10–15% of infertile men. It requires urgent specialist referral to determine the type:
Obstructive azoospermia (OA): Sperm are being produced but cannot reach the ejaculate due to a blockage. Hormone levels (FSH, testosterone) are typically normal. Causes include vasectomy, prior infection, CBAVD, or ejaculatory duct obstruction. Sperm can often be surgically retrieved for IVF/ICSI.
Non-obstructive azoospermia (NOA): The testes are producing very few or no sperm. FSH is often elevated. Causes include genetic conditions (Klinefelter syndrome, Y-chromosome microdeletions), previous chemotherapy or radiation, cryptorchidism (undescended testes), or idiopathic failure. Micro-TESE (microsurgical testicular sperm extraction) may find sperm in approximately 50% of cases.
The distinction between OA and NOA is critical because it determines the treatment pathway. Physical examination, hormonal testing, and genetic testing are all important in making this distinction.
How Often Should You Test?
For Fertility Investigation
NICE guidelines and most fertility specialists recommend at least two semen analyses, collected 2–4 weeks apart, to confirm any abnormal findings. This is because of the significant natural variation between samples.
Some specialists prefer to wait 3 months between tests, as this represents a full cycle of spermatogenesis (sperm production takes approximately 74 days). This is particularly relevant if you've made lifestyle changes or started supplementation—you need at least one full cycle of sperm production to see improvements.
For Monitoring Treatment or Lifestyle Changes
If you've started antioxidant supplementation, made significant lifestyle modifications (quit smoking, lost weight, etc.), or had medical treatment (varicocele repair, hormonal therapy), a follow-up semen analysis after 3–6 months is recommended to assess improvement.
Post-Vasectomy Testing
Post-vasectomy semen analysis follows a different protocol. You'll typically be tested at 12 weeks after the procedure (and after at least 20 ejaculations) to confirm the absence of sperm. A single sample showing azoospermia is usually sufficient, but some clinics request two confirmatory samples.
What to Do With Your Results
If Results Are Normal
✅ Continue trying to conceive naturally—most couples with no identified factors conceive within 2 years
✅ Maintain healthy lifestyle habits (diet, exercise, sleep, stress management)
✅ Ensure your partner has been evaluated if pregnancy hasn't occurred after 12 months
✅ Consider DNA fragmentation testing if unexplained infertility persists
If Results Are Abnormal
Your next steps depend on the severity and pattern of abnormalities:
Mild abnormalities (close to reference values):
✅ Repeat the analysis in 2–4 weeks to confirm
✅ Implement lifestyle modifications: stop smoking, reduce alcohol, achieve healthy weight, eat a Mediterranean-style diet, exercise regularly, avoid heat exposure
✅ Consider evidence-based antioxidant supplementation after discussing with your GP
✅ Retest after 3 months of changes
Moderate to severe abnormalities:
✅ Request GP referral to a urologist or fertility specialist
✅ Expect further investigations: hormonal testing, physical examination, ultrasound, genetic testing (if indicated)
✅ Implement lifestyle changes in parallel with medical investigation
✅ Discuss treatment options with your specialist (medical, surgical, or assisted reproduction)
Azoospermia:
✅ Urgent specialist referral for investigation of obstructive vs non-obstructive cause
✅ Hormonal testing, genetic testing, and specialist imaging
✅ Discussion of sperm retrieval options and assisted reproduction

Frequently Asked Questions
How reliable is a single semen analysis?
A single semen analysis provides useful screening information, but semen quality varies significantly between samples. Studies show that concentration, motility, and morphology can fluctuate by 28–43% between consecutive ejaculates in the same man. This is why at least two analyses are recommended before making clinical decisions. If your first result is abnormal, don't panic—it may not reflect your typical sperm quality.
Can stress affect my results?
Yes, psychological stress can impact semen parameters. Stress hormones (cortisol) may interfere with testosterone production and spermatogenesis. Additionally, the anxiety of producing a sample at a clinic can affect ejaculation and potentially the quality of the sample. If you found the experience very stressful, mention this to your clinician—they may suggest home collection for a repeat test.
My morphology is only 3%—should I be worried?
A morphology of 3% is just below the WHO reference of 4%, and on its own, this is rarely a significant concern. Remember that even fertile men typically have very low morphology scores because the strict criteria are extremely demanding. Morphology is also one of the most variable and subjective measurements in semen analysis. If your other parameters (concentration and motility) are normal, mildly low morphology is unlikely to significantly impair your fertility.
What's the difference between "total motility" and "progressive motility"?
Total motility includes all moving sperm—both those swimming forward (progressive) and those moving but not going anywhere (non-progressive). Progressive motility specifically measures sperm swimming in a forward direction, which is what matters for reaching the egg. Progressive motility is generally considered the more clinically important measure. The WHO 2021 references are ≥42% for total motility and ≥30% for progressive motility.
Does age affect semen analysis results?
Yes, though the decline is more gradual than female fertility decline. Research suggests that sperm concentration remains relatively stable, but motility, morphology, and DNA integrity tend to decrease from the mid-30s onwards. Semen volume may also decrease with age. These changes are generally modest but become more pronounced after age 45–50.
Can medications affect my results?
Yes, several medications can impact sperm production or function. These include testosterone replacement therapy (which paradoxically suppresses natural sperm production), anabolic steroids, certain antidepressants (SSRIs), some blood pressure medications, chemotherapy agents, and 5-alpha-reductase inhibitors (finasteride, dutasteride) used for hair loss or prostate conditions. Always inform the laboratory and your clinician about any medications you're taking.
My GP says my results are "normal" but we still can't conceive—what now?
Standard semen analysis captures only some aspects of sperm function. Several important factors aren't measured in routine testing, including sperm DNA fragmentation, the acrosome reaction (the enzyme release needed to penetrate the egg), and sperm-egg binding capacity. Ask your GP about referral for DNA fragmentation testing, and ensure your partner has had a comprehensive fertility evaluation including assessment of ovulation, tubal patency, and uterine health.
How do I know if I need DNA fragmentation testing?
DNA fragmentation testing is worth considering if you have unexplained infertility (normal semen analysis, no female factors), recurrent miscarriage, repeated IVF/ICSI failure despite good embryo quality, a known varicocele, or if you're over 40. It's also useful if you smoke or have significant oxidative stress risk factors. The test is not routinely available on the NHS but can be accessed privately for approximately £200–£400.
Should I request a specific laboratory or clinic?
Semen analysis quality depends heavily on laboratory expertise. Look for laboratories that perform regular quality control, participate in external quality assessment schemes, and follow WHO methodology. High-volume andrology laboratories (those performing semen analysis regularly) tend to provide more reliable results than general pathology laboratories that analyse semen samples only occasionally.
When to Seek Specialist Help
Don't delay seeking professional evaluation if:
❌ You've been trying to conceive for 12 months without success (6 months if your partner is over 35)
❌ Your semen analysis shows moderate to severe abnormalities on two separate occasions
❌ You have azoospermia (no sperm) on any test
❌ You have known risk factors: undescended testes, prior testicular surgery/injury, varicocele, previous chemotherapy, genetic conditions
❌ You experience sexual dysfunction: erectile difficulties, ejaculation problems, or pain
❌ You notice physical changes: testicular lumps, swelling, pain, or changes in size
Early investigation provides the best outcomes. Male fertility issues are often treatable, and even in complex cases, advances in assisted reproduction mean that fatherhood is achievable for many men who would previously have had no options.
All scientific evidence cited in this article is hyperlinked throughout the text. Click any linked study to access the full research on PubMed.
Related Articles
Prenatal Vitamins UK: What You Actually Need When Trying to Conceive
Male Fertility Supplements UK: Evidence-Based Guide 2026
When to See a Fertility Doctor UK: Guide for Trying to Conceive
Low Sperm Motility: Causes, Treatment & What Actually Helps
Medical Disclaimer This article is for informational and educational purposes only and is not intended as medical advice. Semen analysis results should always be interpreted by a qualified healthcare professional in the context of your individual circumstances. Always consult with your GP or a qualified fertility specialist for personalised guidance. The information provided should not replace professional medical consultation or delay seeking appropriate care. If you have been trying to conceive for over 12 months (or 6 months if your partner is over 35), seek professional evaluation without delay.